| 101. |
Senthilkumar MB, Sarangi P, Amit S, Senguttuvan S, Kumar N, Jayandharan GR*. miR-450b-5p is a key molecular mediator in the development of hemophilic arthropathy. Haemophilia, 2026, in press. |
| 100. |
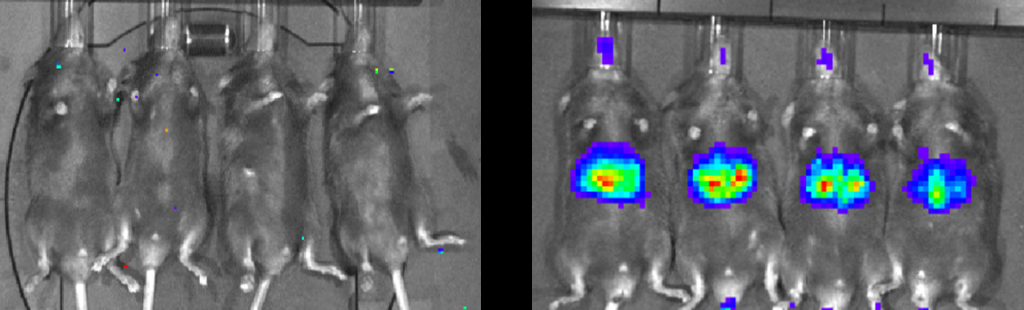
Mohankumar B. S, Sharma S, Srinivasan N, Varghese A, Sarangi P, Singh V, Kumar N, Yenurkar D, Mukherjee S, Amit S, Bhatia S, Misra SK, Puri RD, Chamberlain J, Jayandharan GR*. Bioengineered AAV9 and optimized microdystrophin vectors augment phenotypic rescue in a murine model of Duchenne muscular dystrophy. J Cell Mol Med, 2026, 30:e71078. |
| 99. |
Huda S, Mohankumar BS, Bihani D, Sarangi P, Narendra Kumar, Chandra D, Jayandharan GR*. Optimized AAVrh10 and Factor 9 vectors demonstrate improved phenotypic rescue in hemophilia B mice. Thromb Res, 2026, in press. |
| 98. |
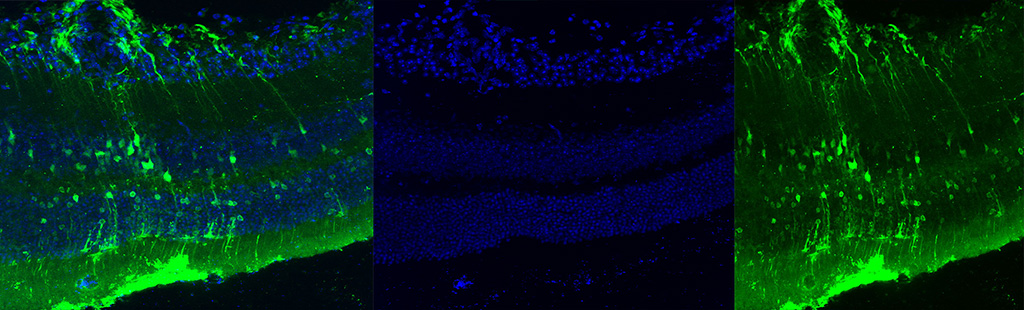
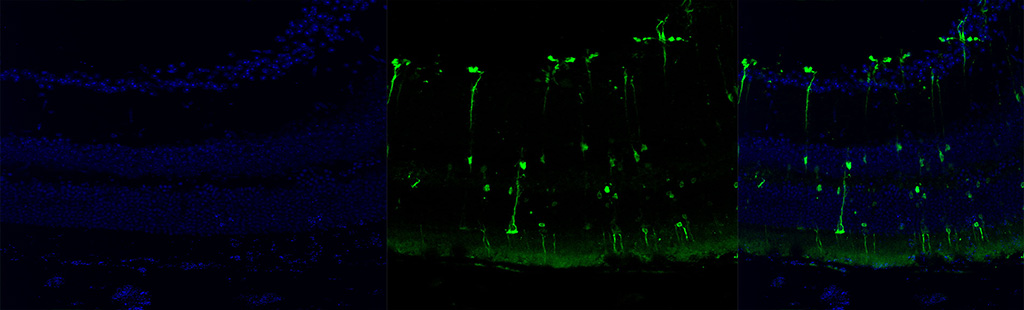
Singh V, Huda S, Kumar N, Jayandharan GR*. Combination gene therapy with AAV based RPE65 and Survivin vectors sustains phenotypic rescue in neural retina of LCA2 mice. Mol Cell Biochem, 2026, in press. |
| 97. |
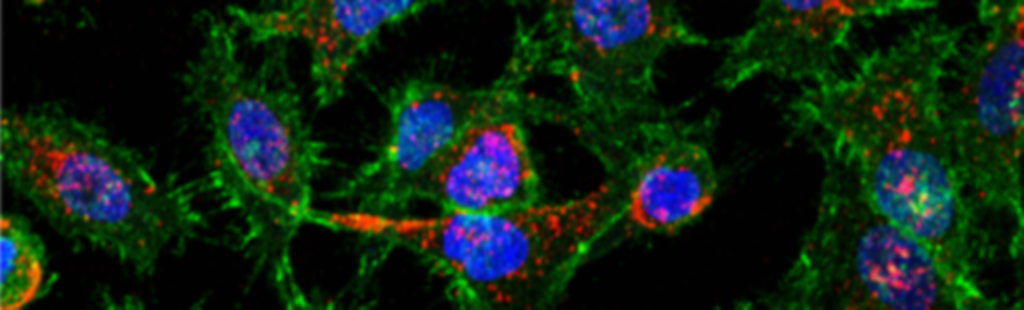
Tripathi S, Huda S, Kar J, Chandra D, Jayandharan GR*, Mohan N*. Suppressing microtubule detyrosination augments AAV2 endosomal escape and gene delivery. J of Cell Sci, 2025, 138(23): jcs264190. |
| 96. |
Pathak S, Singh V, Kumar N, Jayandharan GR*. AAV-mediated combination gene therapy of inducible Caspase 9 and miR-199a-5p is therapeutic in hepatocellular carcinoma. Cancer Gene Ther 2024 31, 1796-1803. |
| 95. |
Singh V, Pathak S, Kumar N, Jayandharan GR*. Development of an Optimized Promoter System for Exosomal and Naked AAV Vector-Based Suicide Gene Therapy in Hepatocellular Carcinoma. ACS Omega 2024, 9(28):30945-30953 |
| 94. |
Sarangi P, Senthilkumar MB, Amit S, Kumar N, Jayandharan GR*. AAV mediated repression of Neat1 lncRNA combined with F8 gene augmentation mitigates pathological mediators of joint disease in haemophilia. J Cell Mol Med. 2024, 28(11):e18460. |
| 93. |
Sarangi P, Kumar K, Sambasivan R, Ramalingam S, Amit S, Chandra D, Giridhara R. Jayandharan* . AAV mediated genome engineering with a bypass coagulation factor alleviates the bleeding phenotype in a murine model of hemophilia B. Thrombosis Res. 2024, 238:151-160. |
| 92. |
Maddileti S, Mahato S, Agrawal T, Dave VP, Naik M, Ali MJ, Kannabiran C, Jalali S, Jayandharan GR, Mariappan I. Generation of two induced pluripotent stem cell lines (LVPEIi004-A and LVPEIi005-B) from probands with Leber Congenital Amaurosis 2 (LCA2) and harboring mutations in RPE65. Stem Cell Res. 2024 3;77:103413. |
| 91. |
Pathak S, Singh V, Kumar N, Jayandharan GR*. Inducible Caspase 9 mediated suicide gene therapy using AAV6 vectors in a murine model of breast cancer. Mol Ther Methods Clin Dev 2023, 31:101166. |
| 90. |
Senthilkumar MB, Sarangi P, Amit S, Senguttuvan S, Kumar N, Jayandharan GR*. Targeted delivery of miR125a-5p and human Factor VIII attenuates molecular mediators of hemophilic arthropathy. Thrombosis Res. 2023,231:8-16. |
| 89. |
Cornetta K*, Kay S*, Urio F, Minja IK, Mbugi E, Mgaya J, Mselle T, Nkya S, Alimohamed MZ, Ndaki K, Bonamino M, Koya RC, Shah LD, Mahlangu J, Drago D, Rangarajan S, Jayandharan GR. Implementation of a Gene Therapy Education Initiative by the ASGCT and Muhimbili University of Health and Allied Sciences. Mol Ther, 2023, 31:2561-2565. |
| 88. |
Sarangi P, Senthilkumar MB, Kumar N, Senguttuvan S, Vasudevan M, Jayandharan GR*. Potential role of long non-coding RNA H19 and Neat1 in haemophilic arthropathy.
J Cell Mol Med. 2023 May 14. doi: 10.1111/jcmm.17770. |
| 87. |
Rajagopal P, Jayandharan GR, Krishnan UM. Polyketal-based nanocarriers: A new class of stimuli-responsive delivery systems for therapeutic applications. European Polymer Journal. 2022, 173: 111290. |
| 86. |
Pathak S, Sarangi P, Jayandharan GR*. Gene therapy for female infertility: A farfetched dream or reality?. Cell Rep Med. 2022, 3(5):100641.
|
| 85. |
Cornetta K, Bonamino M, Mahlangu J, Mingozzi F, Rangarajan S, Rao J. Gene therapy access: global challenges, opportunities and views from Brazil, South Africa, and India. Mol Ther. 2022 doi: 10.1016/j.ymthe.2022.04.002. |
| 84. |
Maurya S, Sarangi P, Jayandharan GR. Safety of Adeno-associated virus-based vector mediated gene therapy- impact of vector dose (Editorial). Cancer Gene Ther 2022, in press. |
| 83. |
Singh V, Khan N, Jayandharan GR. Vector engineering, strategies and targets in cancer gene therapy. Cancer Gene Ther 2021,doi: 10.1038/s41417-021-00331-7. |
| 82. |
Rajagopal P, Jayandharan GR, Krishnan UM. Evaluation of the Anticancer Activity of pH-Sensitive Polyketal Nanoparticles for Acute Myeloid Leukemia. Mol Pharmaceutics, 2021, DOI: 10.1021/acs.molpharmaceut.0c01243. |
| 81. |
Maurya S, Jayandharan GR. Gene Therapy: Contest between Adeno-associated virus and host cells and the impact of UFMylation. Mol Pharmaceutics, 2020, 17:3649-3653. |
| 80. |
Khan N, Maurya S, Bammidi S, Jayandharan GR. AAV6 vexosomes mediate robust suicide gene delivery in a murine model of hepatocellular carcinoma. Mol Ther Methods Clin Dev 2020, 17:497-504. |
| 79. |
Maurya S, Jayandharan GR. Exosome-associated SUMOylation mutant AAV demonstrates improved ocular gene transfer efficiency in vivo Virus Res 2020, in 283:197966. |
| 78. |
Maurya S, Mary B, Jayandharan GR. Improved ocular gene transfer with a Neddylation-site modified AAV-RPE65 vector in rd12 mice. Eye 2020, doi:10.1038/s41433-020-0838-8. |
| 77. |
Khan N#, Sabna C#, Saxena H#, Bammidi S, Jayandharan GR. Development of a small RNA based recombinant AAV vector production method to improve the potency of suicide gene therapy vectors. Cancer Med 2020, doi: 10.1002/cam4.2935. (#equal contribution). |
| 76. |
Arumugam S#, Mary B#, Kumar M, Jayandharan GR. (#equal contribution). Analysis of hepatic and retinal cell microRNAome during AAV infection reveals their diverse impact on viral transduction and cellular physiology.Gene 2020, 724:144157. |
| 75. |
Maurya S, Mary B, Jayandharan GR. Rational engineering and pre-clinical evaluation of Neddylation and SUMOylation site modified AAV vectors in murine models of hemophilia B and Leber congenital amarousis. Hum Gene Ther 2019, 30:1461-1476. |
| 74. |
Mary B, Maurya S, Kumar M, Bammidi S, Kumar V, Jayandharan GR. Molecular engineering of Adeno-associated virus capsid improves its therapeutic gene transfer in murine models of hemophilia and retinal degeneration. Molecular Pharmaceutics 2019, 16(11):4738-4750. |
| 73. |
Khan N, Bammidi, S, Jayandharan, GR. A CD33 antigen targeted AAV6 vector expressing an inducible caspase-9 suicide gene is therapeutic in a xenotransplantation model of acute myeloid leukemia. Bioconjugate Chem 2019, 30:2404-2416. |
| 72. |
Purohit N, Jain A, Mathews V, Jayandharan GR. Molecular characterization of novel Adeno-associated virus variants infecting human tissues. Virus Research 2019, 13; 272:197716. |
| 71. |
Mary B, Maurya S, Arumugam S, Kumar V, Jayandharan GR. Post-translational modifications in capsid proteins of recombinant adeno-associated virus (AAV) 1-rh10 serotypes. FEBS J. 2019, 286:4964-498. |
| 70. |
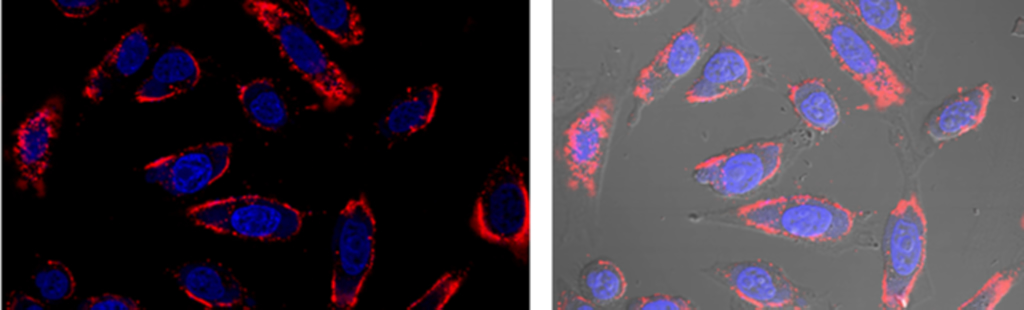
Mary B, Jayandharan GR. Visualization of retinal morphology in rd12mice by scanning electron microscopy. Hum Gene Ther. 2019 doi:10.1089/hum.2019.116. |
| 69. |
Khan N, Bammidi S, Chattopadhyay S, Jayandharan GR. Combination suicide gene delivery with an AAV vector encoding inducible caspase-9 and a chemical inducer of dimerization is effective in a xenotransplantation model of hepatocellular carcinoma. Bioconjugate Chem 2019, 30:1754-1762. |
| 68. |
Khan N, Mahajan NK, Sinha P, Jayandharan GR. An efficient method to generate xenograft tumor models of acute myeloid leukemia and hepatocellular carcinoma in adult zebrafish. Blood Cell Mol Dis 2019,75: 48-55. |
| 67. |
Mary B, Khan N, Arumugam S, Saxena H, Kumar M, Manimaran P, Chattopadhyay S, Jayandharan GR. Adeno-associated Virus Vectors in Gene Therapy. In: Jayandharan G. (eds) Gene and Cell Therapy: Biology and Applications. 2018, Springer, Singapore |
| 66. |
Mahato M, Jayandharan GR, Vemula PK. Viral- and Non-viral-Based Hybrid Vectors for Gene Therapy. In: Jayandharan G. (eds) Gene and Cell Therapy: Biology and Applications. 2018, Springer, Singapore |
| 65. |
Rajasekaran S, Thatte J, Periasamy J, Javali A, Jayaram M, Sen D, Krishnagopal A, Jayandharan GR, Sambasivan R. Infectivity of adeno-associated virus serotypes in mouse testis. BMC Biotechnology, 2018. 18: 70 BMC https://doi.org/10.1186/s12896-018-0479-1 |
| 64. |
Rajagopal P, Duraiswamy S, Sethuraman S, Jayandharan GR, Krishnan UM. Polymer-coated viral vectors: hybrid nanosystems for gene therapy. J Gene Med, 2018, 20: e3011 |
| 63. |
Selot R, Arumugam S, Mary B, Cheemadan S and Jayandharan GR.Optimized AAV rh.10 vectors that partially evade neutralizing antibodies during hepatic gene transfer. Front. Pharmacol. 2017; doi: 10.3389/fphar.2017.00441 |
| 62. |
Gabriel N, Samuel R, Jayandharan GR.Targeted delivery of adeno associated virus transduced mesenchymalstromal cells to hepatic tissue for ex vivo gene delivery. J Tissue EngRegen Med;2017, 11 :1354-1364. |
| 61. |
Balakrishnan B, Jayandharan GR. Intra-cellular trafficking of AAV5 vectors. Hum Gene Ther Methods 2016; 27: 47-8. |
60. |
Hareendran S, Ramakrishna B, Jayandharan GR. Synergistic inhibition of PARP-1 and NF-kB signaling down-regulates immune response against recombinant AAV2 vectors during hepatic gene therapy. Eur J Immunol 2016, 46:154-66. |
| 59. |
Selot R, Marepally S, Vemula PK, Jayandharan GR. Nanoparticle coated viral vectors for gene therapy. Curr Biotech, 2016, 5: 44 – 53. |
| 58. |
Arumugam S. Jayandharan GR. Adeno-associated virus (AAV)-2 Genome in Arthrobacter sp. LS16? J Genet Syn Gene Ther 2016, 7:2. |
| 57. |
Sen D, Jayandharan GR. microRNA-15b modulates molecular mediators of blood induced arthropathy in hemophilia mice. Int J Mol Biosci 2016, 17: 492. doi:10.3390/ijms17040492. |
| 56. |
Rao R, Dhele N, Cheemadan S, Ketkar A, Jayandharan GR , Palakodeti D and Rampalli S. H3K27me3 activity favors somatic transition during human pluripotent reprogramming. Sci Rep; 2015, 5: 8229 | DOI: 10.1038/srep08229. |
| 55. |
Ling C, Wang Y, Lu Y, Wang L, Jayandharan G, Aslanidi G, Li B, Cheng B, Ma W, Lentz T, Ling C, Xiao X, Samulski R, Muzyczka N, Srivastava A. Enhanced transgene expression from recombinant single-stranded D-sequence−substituted AAV vectors in human cell lines in vitro and in murine hepatocytes in vivo. J Virol 2015; 89: 952-61. |
| 54. |
Ling C, Wang Y, Lu Y, Wang N, Jayandharan GR, Aslanidi GV, Li B, Cheng B, Ma W, Lentz T, Ling C, Xiao X, Samulski RJ, Muzyczka N and Srivastava A-The Adeno-Associated Virus Genome Packaging Puzzle. J Mol Genet Med 2015, 9:178. doi:10.4172/1747-0862.1000178. |
| 53. |
Sen D, Balakrishnan B, Jayandharan GR. Cellular unfolded protein response against viruses used in gene therapy. Front Microbiol 2014; 5:250. |
| 52. |
Senapati J, Devasia AJ, David S, Manipadam MT, Nair S, Jayandharan GR, George B. Diffuse large B cell lymphoma in wiskott-Aldrich syndrome: a case report and review of literature. Indian J Hematol Blood Transfus. 2014; 30:309-13. |
| 51. |
Selot R, Hareendran S, Jayandharan GR. Developing immunologically inert adeno-associated virus (AAV) vectors for gene therapy: possibilities and limitations. Curr Phar Biotech 2014;14:1072-82. |
| 50. |
Balakrishnan B, Jayandharan GR. Basic biology of Adeno-associated virus (AAV) vectors used in gene therapy. Curr Gene Ther 2014;14:86-100. |
| 49. |
Batchu RB, Gruzdyn OV, Moreno-Bost AM, Szmania S, Jayandharan G, Srivastava A,Kolli BK, Weaver DW, van Rhee F, Gruber SA. Efficient lysis of epithelial ovariancancer cells by MAGE-A3-induced cytotoxic T lymphocytes using rAAV-6 capsidmutant vector. Vaccine 2014; 32:938-43.
|
| 48. |
Sen D, Balakrishnan B, Gabriel N, Agrawal P, Roshini V, Samuel R, Srivastava A, Jayandharan GR. Improved adeno-associated virus (AAV) serotype 1 and 5 vectors for gene therapy. Sci Rep; 2013, 3: 1832. DOI: 10.1038/srep01832. |
| 47. |
Sangeetha H, Balakrishnan B, Sen D, Kumar S, Srivastava A, Jayandharan GR. Adeno-associated virus (AAV) vectors in gene therapy: Immune challenges and strategies to circumvent them. Rev Med Virol; 23:399-413. |
| 46. |
Gabriel N, Hareendran S, Sen D, Gadkari RA, Govindarajan S, Hussain M, Duraiswamy R, Samuel R, Srinivasan N, Srivastava A, Jayandharan GR. Bio-Engineering of Adeno-Associated Virus Serotype (AAV)-2 Capsid at Serine/Threonine/Lysine Residues Improves Its Transduction Efficiency Both In Vitro and In Vivo. Hum Gene Ther Methods 2013; 24: 80-93. |
| 45. |
Sen D, Gadkari RA, Sudha G, Gabriel N, Sathish Kumar Y, Selot R, Samuel R, Rajalingam S, Ramya V, Nair SC, Srinivasan N, Srivastava A, Jayandharan GR.Targeted modifications in adeno-associated virus (AAV) serotype -8 capsid improves its hepatic gene transfer efficiency in vivo. Hum Gene Ther Methods 2013; 24:104-16. |
| 44. |
Balakrishnan B, Sen D, David S, Srivastava A, Srivastava A, Jayandharan GR. Activation of the Cellular Unfolded Protein Response by Recombinant Adeno-Associated Virus Vectors. PLoS ONE 2013; 8(1): e53845. doi:10.1371/journal.pone.0053845. |
| 43. |
Sen D, Chapla A, Walter N, Daniel V, Srivastava A, Jayandharan GR. Nuclear Factor (NF)-κB is a major regulator of blood induced joint damage in a murine model of hemophilia. J Thromb Haemost 2013, 11: 293-306. |
| 42. |
Devi GS, Arora N, Rajkumar SV, Edison ES, Srivastava A, Jayandharan GR. Fluorescent PCR-based gene dose analysis for detection of deletion mutations in carriers of haemophilia. Haemophilia. 2013 Jul 16. doi: 10.1111/hae.12233. |
| 41. |
Jain R, T L, Chandran J, Jayandharan GR, Palle A, Moses PD. A novel mutation c.1048A>T at codon 350(Lys>Stop) in PROC gene causing neonatal purpura fulminans. Blood Coagul Fibrinolysis. 2013; 24:890-2. |
| 40. |
Song L, Li X, Jayandharan GR, Wang Y, Aslanidi GV, Ling C, Zhong L, Gao G, Yoder MC, Ling C, Tan M, Srivastava A. High-efficiency transduction of primary human hematopoietic stem cells and erythroid lineage-restricted expression by optimized AAV6 serotype vectors in vitro and in a murine xenograft model in vivo. PLoS ONE 2013; 8(3):e58757. |
| 39. |
Sumitha E, Jayandharan GR, Arora N, Abraham A, David S, Sankari Devi G, ShenbagaPriya P, Nair SC, George B, Mathews V, Chandy M, Viswabandya A, Srivastava A. Molecular basis of quantitative fibrinogen disorders in 27 patients from India. Haemophilia 2013, 19: 611-8. |
| 38. |
Song L, Kauss MA, Kopin E, Chandra M, Ul-Hasan T, Miller E, Jayandharan GR, Rivers AE, Aslanidi GV, Ling C, Li B, Ma W, Li X, Andino LA, Zhong L, Tarantal AF, Yoder MC, Wong KK, Jr, Tan M, Chatterjee S, Srivastava A. Optimizing the transduction efficiency of human hematopoietic stem cells using capsid-modified AAV6 vectors in vitro and in a xenograft mouse model in vivo. Cytotherapy 2013,15(8):986-998. |
| 37. |
Jayandharan GR, Srivastava A, Srivastava A. Application of molecular genetics in haemophilia: from diagnoses to therapy. Sem Thromb Haemost 2012; 38: 64-78. |
36. |
Goodeve AC, Perry DJ, Cumming T, Hill M, Jennings I, Kitchen S, Walker I, Gray E, Jayandharan G, Tuddenham E. Genetics of Haemostasis. Haemophilia 2012; 18: S4, 73-80. |
| 35. |
Jayandharan GR, Srivastava A. Hemophilia: disease, diagnosis and management. J Genet Syndr Gene Ther; 2012. doi: 10.4172/2157-7412.S1-005. |
| 34. |
Zhong L, Jayandharan GR, Aslanidi G, Zolutukin S, Herzog RW, Srivastava A. Development of Novel Recombinant AAV Vectors and Strategies for the Potential Gene Therapy of Hemophilia J Genet Syndr Gene Ther; 2012. doi: 10.4172/2157-7412.S1-008. |
| 33. |
David S, Jayandharan GR, Abraham A, Rr J, Devi GS, Patkar N, Shaji RV, Nair SC, Viswabandya A, Ahmed R, George B, Mathews V, Chandy M, Srivastava A. Molecular basis of Wiskott-Aldrich syndrome in patients from India. Eur J Haematol. 2012 doi: 10.1111/j.1600-0609.2012.01818. |
| 32. |
Aslanidi GV, Rivers AE, Ortiz L, Govindasamy L, Ling C, Jayandharan GR, Zolotukhin S, Agbandje-McKenna M, Srivastava A. High-efficiency transduction ofhuman monocyte-derived dendritic cells by capsid-modified recombinant AAV2 vectors. Vaccine. 2012; 30: 3908-17. |
| 31. |
Rogers GL, Martino AT, Aslanidi G, Jayandharan GR, Srivastava A, Herzog RW. Innate immune responses to AAV vectors. Front Microbiol, 2011; 2: e194. |
| 30. |
Sumitha E, Jayandharan GR, David S, Jacob RR, Sankari Devi G, Nair SC, George B, Viswabandya A, Mathews V, Chandy M, Srivastava A. Molecular basis of Bernard Soulier syndrome in 27 unrelated patients in India: Identification of four common ancestral mutations and their genotype-phenotype correlations. J Thromb Haemost 2011; 9: 1590-8. |
| 29. |
Chapla A, Jayandharan GR, Sumitha E, Sankari Devi G, Nair SC, George B, Mathews V, Chandy M, Srivastava A. Genotype-phenotype correlation of hereditary factor V deficiency in India: Thromb Haemost 2011; 105:1120-3. |
| 28. |
Ma W, Li B, Ling C, Jayandharan GR, Srivastava A, Byrne BJ. A simple method to increase the transduction efficiency of single-stranded AAV vectors in vitro and in vivo. Hum Gene Ther 2011; 22: 633-40. |
| 27. |
Petrs-Silva H, Dinculescu A, Li Q, Deng WT, Pang JJ, Min SH, Chiodo V, Neeley AW, Govindasamy L, Bennett A, Agbandje-McKenna M, Zhong L, Li B, Jayandharan GR, Srivastava A, Lewin AS, Hauswirth WW. Novel Properties of Tyrosine-mutant AAV2 Vectors in the Mouse Retina. Mol Ther 2011; 19: 293-301. |
| 26. |
Jayandharan GR, Zhong L, Sack BK, Rivers AE, Li M, Li B, Herzog RW, Srivastava A. Optimized AAV-protein phosphatase 5 helper-viruses for efficient transduction by single-stranded AAV vectors: Therapeutic expression of Factor IX at reduced vector doses. Hum Gene Ther 2010; 21: 271-83. |
| 25. |
Preston FE, Lippi G, Favaloro EJ, Jayandharan GR, Edison ES, Srivastava A. Quality issues in laboratory haemostasis. Haemophilia 2010; Suppl 5:93-9. |
| 24. |
Markusic DM, Herzog RW, Aslanidi GV, Hoffman BE, Li B, Li M, Jayandharan GR, Ling C, Zolotukhin I, Ma W, Zolotukhin S, Srivastava A, Zhong L. High-efficiency transduction and correction of murine hemophilia B using AAV2 vectors devoid of multiple surface-exposed tyrosines. Mol Ther 2010; 18: 2048-56. |
| 23. |
Ling C, Lu Y, Kelsi JK, Jayandharan GR, Li B, Ma W, Cheng B, Gee SW, McGoogan KE, Govindasamy L, Agbandje-McKenna M, Zhong L, Srivastava A. Human Hepatocyte Growth Factor Receptor is a cellular Co-Receptor for AAV3. Hum Gene Ther 2010; 21:1741-7. |
| 22. |
Qiao C, Zhang W, Yuan Z, Shin JH, Li J, Jayandharan GR, Zhong L, Srivastava A, Xiao X, Duan D. AAV6 capsid tyrosine to phenylalanine mutations improve gene transfer to skeletal muscle. Hum Gene Ther 2010; 21: 1343-8. |
| 21. |
Li M, Jayandharan GR, Li B, Chen L, Ma W, Srivastava A, Zhong L.
High-Efficiency transduction of fibroblasts and mesenchymal Stem Cells by tyrosine-Mutant AAV2 vectors for their potential use in cellular therapy. Hum Gene Ther 2010; 21: 1527-43. |